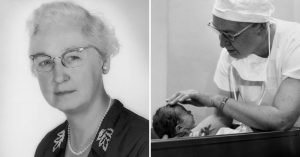
Dr. Frances Kelsey resisted intense industry pressure to approve thalidomide; the drug was the cause of severe birth defects in over 10,000 infants in other countries.
 When pharmacologist Dr. Frances Oldham Kelsey started working at the U.S. Food and Drug Administration (FDA) in 1960, one of the first files to cross her desk was an approval request for thalidomide. The drug had already been prescribed widely in Europe and other countries as a treatment for morning sickness in pregnant women, but Kelsey wasn't convinced it was safe. Her refusal to approve the drug, despite intense pressure from its manufacturer, likely saved tens of thousands of babies in America from devastating birth defects. "Representatives for the company thought I was crazy because it was such a popular drug in Europe, and they were losing money by my pigheadedness," asserted Kelsey in a later interview. "I held my ground. I just wouldn't approve it."
When pharmacologist Dr. Frances Oldham Kelsey started working at the U.S. Food and Drug Administration (FDA) in 1960, one of the first files to cross her desk was an approval request for thalidomide. The drug had already been prescribed widely in Europe and other countries as a treatment for morning sickness in pregnant women, but Kelsey wasn't convinced it was safe. Her refusal to approve the drug, despite intense pressure from its manufacturer, likely saved tens of thousands of babies in America from devastating birth defects. "Representatives for the company thought I was crazy because it was such a popular drug in Europe, and they were losing money by my pigheadedness," asserted Kelsey in a later interview. "I held my ground. I just wouldn't approve it."
Born in Cobble Hill, British Columbia on July 24, 1914, Kelsey's early love of science led her to study pharmacology and she earned bachelor's and master's degrees at McGill University. After graduating in 1935, a professor encouraged her to contact the noted researcher Dr. Eugene Geiling, who was setting up a new pharmacology department at the University of Chicago. Geiling offered her a research position — but the letter was addressed to Mr. Oldham. "My conscience tweaked me a bit," she wrote later. "Should I write and explain that Frances with an 'e' is female and with an 'i' is male? [My supervisor] said, 'Don't be ridiculous. Accept the job, sign your name, put Miss in brackets afterwards, and go!'" Still, she often thought about that mistake: "to this day, I do not know if my name had been Elizabeth or Mary Jane, whether I would have gotten that first big step up."
"Representatives for the company thought I was crazy because it was such a popular drug in Europe, and they were losing money by my pigheadedness... I held my ground. I just wouldn't approve it."
In Chicago, Kelsey had her first experience working with the FDA, assisting on a research project that showed that 107 deaths of people who had taken a sulfonamide were caused by use of an improper solvent in the medication's manufacture. In 1938, she received a PhD in pharmacology at the University of Chicago and joined the faculty. While researching cures for malaria, she discovered that some medications could cross the placental barrier in pregnant women. She continued her studies and was awarded her MD in 1950.
In 1960, after becoming a dual citizen of the U.S. and Canada, Kelsey was hired by the FDA in Washington, DC as one of only seven full-time employees and four part-time physicians examining drug approvals. When she started working at the FDA, she was part of a new generation of scientists who wanted to ensure that drugs were proven safe and effective before being marketed to the public. At the time, the FDA did not have the authority to enforce these requirements and pharmaceutical companies often engaged in minimal safety testing. It was even a common practice for companies to send new or experimental drugs to doctors for them to try out on their patients.
 During her first month at her new job, one of Kelsey's first assignments was to review the application from the Richardson-Merrell Company for approval of thalidomide. The company expected a fast approval since the drug was already in use in numerous countries including Germany, the UK, Canada, and Australia. Distrustful of the information provided by the company and, as she later explained, feeling that its representatives were "at no time being wholly frank with me" about the drug's safety, Kelsey told the company further tests were needed. Over several months, the company continued to submit their application and Kelsey continued to reject it citing the need for additional tests.
During her first month at her new job, one of Kelsey's first assignments was to review the application from the Richardson-Merrell Company for approval of thalidomide. The company expected a fast approval since the drug was already in use in numerous countries including Germany, the UK, Canada, and Australia. Distrustful of the information provided by the company and, as she later explained, feeling that its representatives were "at no time being wholly frank with me" about the drug's safety, Kelsey told the company further tests were needed. Over several months, the company continued to submit their application and Kelsey continued to reject it citing the need for additional tests.
Throughout this period, the company complained to her supervisors, contacted her repeatedly, and, as written in a 1962 Washington Post story, she "[lived] the while with insinuations that she was a bureaucratic nitpicker, unreasonable — even, she said, stupid." Kelsey's determined stance was vindicated eight months after the company's initial filing when evidence from Europe showed that thalidomide was the cause of severe birth defects including children born with missing limbs or flipper-like limbs, malformations of the heart, and deafness and blindness. In Germany alone, between 5,000 and 7,000 children were affected, of which only 40% survived, and thousands of cases emerged in other countries. In the US, fewer than 100 babies were born with thalidomide-related birth defects; the drug was never licensed in the US due to Kelsey's efforts however a small number of pregnant women were exposed to it through samples provided to doctors from the drug company.
After news of the thalidomide disaster broke, there was huge public outcry for better drug testing and the U.S. Congress unanimously passed the Kefauver Harris Amendment in 1962 to strengthen drug regulations and gave the FDA enforcement authority. Kelsey was selected to help write the rules governing clinical trials — rules that have since been adopted throughout the industrialized world — and then oversee their enforcement as the director of the FDA's Investigational Drug Branch.
On August 7, 1962, Kelsey became the second woman ever to receive the country's highest civilian honor when President John F. Kennedy awarded her the President's Award for Distinguished Federal Civilian Service. Kelsey's far-reaching impact on science and drug testing was described by Harvard historian Daniel Carpenter as follows: "She had a huge effect on the science that we all take for granted today... She and the F.D.A. had a huge role in determining the terms and sequence of what is now modern clinical science."
Kelsey retired from the FDA at the age of 90 in 2005 after 45 years of service. In 2010, the FDA presented her with the first Drug Safety Excellence Award and then named it in her honor; announcing that the Dr. Frances O. Kelsey Award had been established to "celebrate courage and scientific decision-making." This determined scientist, whose courageous actions helped save the lives of countless people, passed away in 2015 at the age of 101.
Books And Resources About Women in Science
Charlotte the Scientist Finds A Cure
Charlotte the Scientist Finds A Cure
Creatures all over the forest are getting sick, and Charlotte the bunny scientist is determined to figure out why! The stumped doctors and scientists are dismissive of her efforts, but she holds firm to her beloved grandfather's assertion that she will "make a real difference in the world." After some patient interviews and a few samples from the outhouse, Charlotte realizes that all the sick animals have been munching on carrots contaminated by 'Funky Forest Fungi.' A quick clinical trial later, and Charlotte has saved the tummies of all her friends! This delightful sequel to Charlotte the Scientist Is Squished celebrates the ability of determined girls to change the world.
Dr. Jo: How Sara Josephine Baker Saved the Lives of America's Children
Dr. Jo: How Sara Josephine Baker Saved the Lives of America's Children
After Sara Josephine Baker lost her brother and father to typhoid fever, she knew she wanted to be a doctor. But when she graduated in 1898, few people wanted to see a woman doctor, so Dr. Jo took a job in public health working in Hell's Kitchen, one of New York's poorest neighborhoods. She realized that, by improving the health of children, she could improve the health of a whole community. Dr. Jo assigned visiting nurses to new mothers, designed safe infant clothing, set up milk stations, and created training and licensing for midwives — and her work saved over 90,000 children. This picture book biography of a groundbreaking woman in medicine highlights how simple innovations can have an enormous impact.
Rachel Carson and Her Book that Changed the World
Rachel Carson and Her Book that Changed the World
As a shy young woman, Rachel Carson found joy and purpose in studying the creatures all around her. Her articles and books about marine life made her a best-selling author, but it was her groundbreaking book Silent Spring, about the effects of the pesticide DDT on wild animals and birds, that turned her into a household name — and helped found the modern environmental movement. Written for the fiftieth anniversary of Silent Spring's publication, this thoughtful picture book shows how a quiet and dedicated scientist changed the way people thought of their effects on the Earth. For two more picture books about Carson, check out Rachel Carson: Preserving a Sense of Wonder and Rachel: The Story of Rachel Carson.
Born Just Right
Born Just Right
If you saw the video of a girl using a prosthetic arm that shoots glitter, then you already know about Jordan Reeves! Now, with the help of her mother, Jen Lee Reeves, Jordan gets to tell the story of growing up with a congenital limb difference — and how our attitudes towards disability can either limit kids or set them free. Jordan's frank and funny stories about everything from learning to tie her shoes with a "little arm" to trying every sport she can will intrigue young readers, and her foundation Born Just Right will show them that you're never to young to make a change for the better!
Women in Science
50 Fearless Pioneers Who Changed the World
Women in Science
50 Fearless Pioneers Who Changed the World
This charmingly illustrated and educational book highlights the contributions of fifty notable women to the fields of science, technology, engineering, and mathematics from the ancient to the modern world. Full of striking, singular art, this fascinating collection profiles well-known figures like primatologist Jane Goodall, as well as lesser-known pioneers such as Katherine Johnson, the African-American physicist and mathematician who calculated the trajectory of the 1969 Apollo 11 mission to the moon. Women in Science celebrates the achievements of the intrepid women who have paved the way for the next generation of female engineers, biologists, mathematicians, doctors, astronauts, physicists, and more!
Trailblazers
33 Women in Science Who Changed the World
Trailblazers
33 Women in Science Who Changed the World
Middle grade kids are eager to learn more about the people who have changed our world — so they're the perfect age to introduce to this inspiring collection of influential women in science! Rachel Swaby, the author of the bestselling science book for teen and adult readers, Headstrong: 52 Women Who Changed Science – And The World, profiles 33 women in this book, including groundbreaking figures like Virginia Apgar, Stephanie Kwolek, Sally Ride, and Rachel Carson. Best of all, Swaby emphasizes that it takes work, practice, and determination to become a brilliant scientist, encouraging kids to foster their curiosity about the world and pursue their love of science.
Bold Women of Medicine
21 Stories of Astounding Discoveries, Daring Surgeries, and Healing Breakthroughs
Bold Women of Medicine
21 Stories of Astounding Discoveries, Daring Surgeries, and Healing Breakthroughs
From 19th century battlefield nurses Clara Barton and Florence Nightingale, to modern medical miracle workers like Dr. Catherine Hamlin and Dr. Bonnie Simpson Mason, women have long played a key role in medicine — but their contributions have often been downplayed or forgotten. In this fascinating new title from the Women of Action series, readers get to meet some of the daring and trailblazing women of the past two hundred years of medicine. These women defied prejudices and expectations, created incredible new procedures, and devoted their lives to healing people around the world.
Headstrong: 52 Women Who Changed Science – And The World
Headstrong: 52 Women Who Changed Science – And The World
Learn the often neglected stories of women in science with these 52 engaging capsule biographies! Spanning centuries of courageous thinkers, author Rachel Swaby celebrates women whose specializations range from biology to physics to engineering to programming, from famous names like Sally Ride and Ada Lovelace to lesser-known women like Stephanie Kwolek and Chien-Shiung Wu. While each individual's biography runs for only a few pages, Swaby has done an impressive job of conveying the essence of each scientist's life and work into the profiles, while her light tone urges readers to learn more about each of these groundbreaking women.
100 More Canadian Heroines: Famous and Forgotten Faces
100 More Canadian Heroines: Famous and Forgotten Faces
Teens can read about Kelsey and 99 other amazing Canadian women in this book from author and Canadian historian Merna Forster! Capsule biographies of women in a wide variety of fields capture their spirits of individuality and determination -- the same spirit that sustained Kelsey as she resisted the pressure by Merrell executives to approve thalidomide. With a clear and accessible summary of Kelsey's work, as well as those of so many other women, this title is sure to fascinate young adult readers.
Frankie: How One Woman Prevented a Pharmaceutical Disaster
Frankie: How One Woman Prevented a Pharmaceutical Disaster
When Frances Kelsey refused to approve thalidomide for use in the United States, the drug's manufacturer thought she was being ridiculous: it was a wonder drug, already being used to help women in Europe and Canada who struggled with morning sickness. But when the horrifying consequences of using the drug came to light, Kelsey went from obstructionist bureaucrat to national hero. In the first ever biography of this determined woman, readers get a powerful portrait of a groundbreaking woman whose quiet courage saved untold people.
Periodic Table Building Blocks
Periodic Table Building Blocks
Elements are the building blocks of the universe — and now they can be the building blocks of your toy box with this fun set from Uncle Goose! 20 solid wood building blocks, made in the US from sustainable Michigan basswood, feature the elements of the periodic table painted in non-toxic, child-safe inks. And even though they're building blocks, don't think they're just for babies — chemistry geeks will love these colorful blocks as much at 36 years as 36 months! For two more of Uncle Goose's science-themed block sets, check out the Dinosaur Blocks and the Fossil Blocks.
Chemistry C500
Chemistry C500
If you're interested in learning chemistry, this kit from Thames & Kosmos is a great place to start! The C500 chemistry kit introduces seven central areas of chemistry through 30 classic experiments. Kids will learn about reactions between solids, liquids, and gases; find out about acids and bases; and try some electrochemistry with metals and salts. Almost everything you need is included in this set, including a full-color experiment guide that lets her know how the chemical principles work in practice.