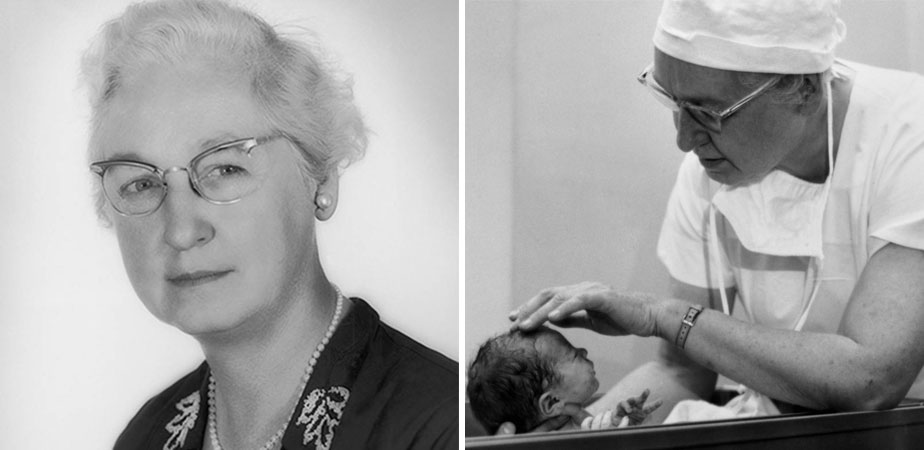
Dr. Frances Kelsey resisted intense industry pressure to approve thalidomide; the drug was the cause of severe birth defects in over 10,000 infants in other countries.
 When pharmacologist Dr. Frances Oldham Kelsey started working at the U.S. Food and Drug Administration (FDA) in 1960, one of the first files to cross her desk was an approval request for thalidomide. The drug had already been prescribed widely in Europe and other countries as a treatment for morning sickness in pregnant women, but Kelsey wasn't convinced it was safe. Her refusal to approve the drug, despite intense pressure from its manufacturer, likely saved tens of thousands of babies in America from devastating birth defects. "Representatives for the company thought I was crazy because it was such a popular drug in Europe, and they were losing money by my pigheadedness," asserted Kelsey in a later interview. "I held my ground. I just wouldn't approve it." Continue reading Continue reading
When pharmacologist Dr. Frances Oldham Kelsey started working at the U.S. Food and Drug Administration (FDA) in 1960, one of the first files to cross her desk was an approval request for thalidomide. The drug had already been prescribed widely in Europe and other countries as a treatment for morning sickness in pregnant women, but Kelsey wasn't convinced it was safe. Her refusal to approve the drug, despite intense pressure from its manufacturer, likely saved tens of thousands of babies in America from devastating birth defects. "Representatives for the company thought I was crazy because it was such a popular drug in Europe, and they were losing money by my pigheadedness," asserted Kelsey in a later interview. "I held my ground. I just wouldn't approve it." Continue reading Continue reading